Atrial and Brain Natriuretic Peptides
Natriuretic peptide formation and release
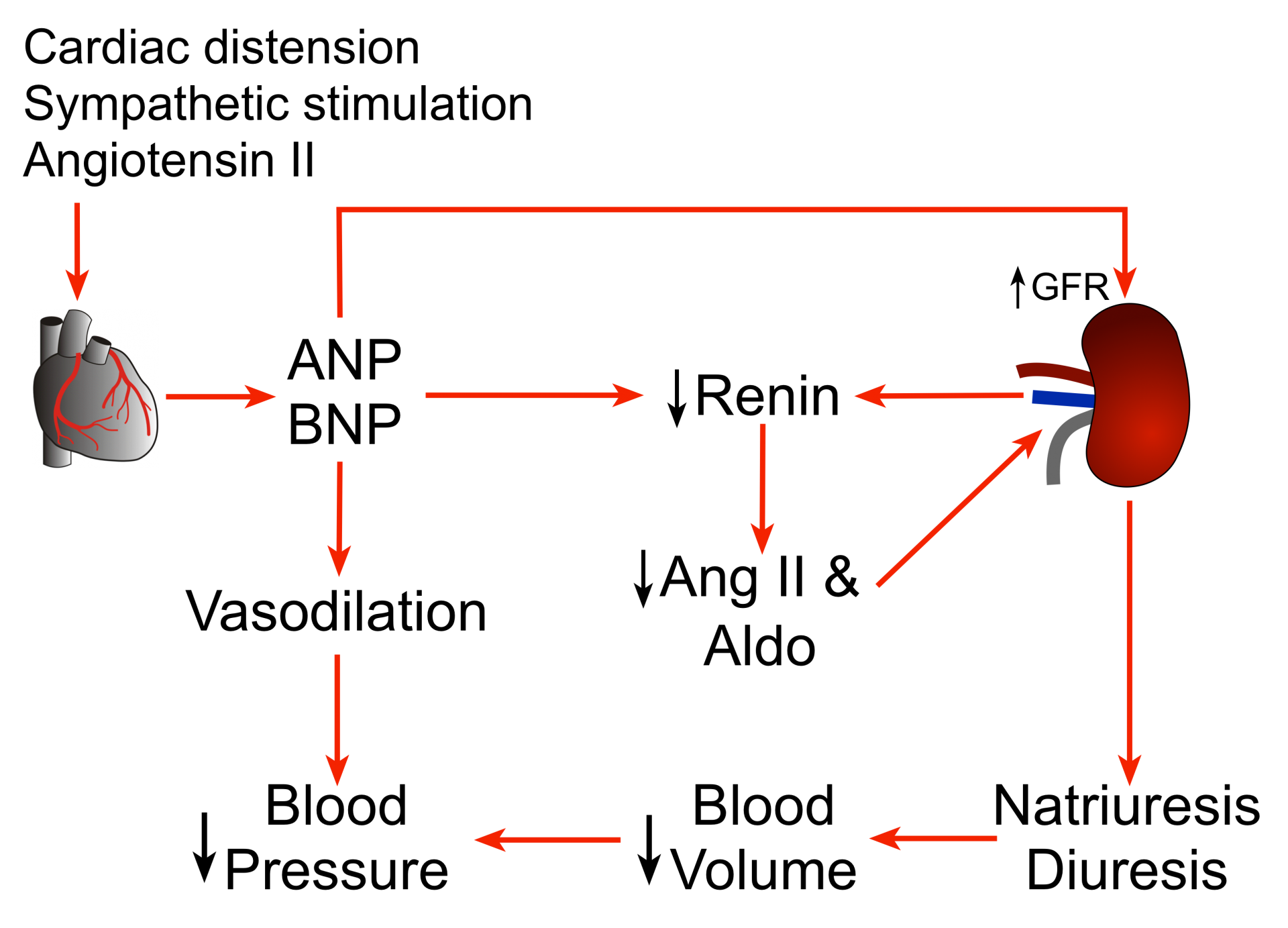 Natriuretic peptides (NPs) are peptide hormones that are synthesized by the heart, brain, and other organs. The release of these peptides by the heart is stimulated by atrial and ventricular distension, as well as by neurohumoral stimuli, usually in response to heart failure. The main physiological actions of natriuretic peptides are to reduce arterial pressure by decreasing blood volume and systemic vascular resistance.
Natriuretic peptides (NPs) are peptide hormones that are synthesized by the heart, brain, and other organs. The release of these peptides by the heart is stimulated by atrial and ventricular distension, as well as by neurohumoral stimuli, usually in response to heart failure. The main physiological actions of natriuretic peptides are to reduce arterial pressure by decreasing blood volume and systemic vascular resistance.
Atrial natriuretic peptide (ANP) is a 28-amino acid peptide that is synthesized, stored, and released by atrial myocytes in response to atrial distension, angiotensin II stimulation, endothelin, and sympathetic stimulation (beta-adrenoceptor mediated). Therefore, elevated levels of ANP are found during hypervolemic states (elevated blood volume), such as occur in heart failure. ANP is first synthesized and stored in cardiac myocytes as prepro-ANP, which is then cleaved to pro-ANP and finally to ANP. ANP is the biologically active form of the peptide.
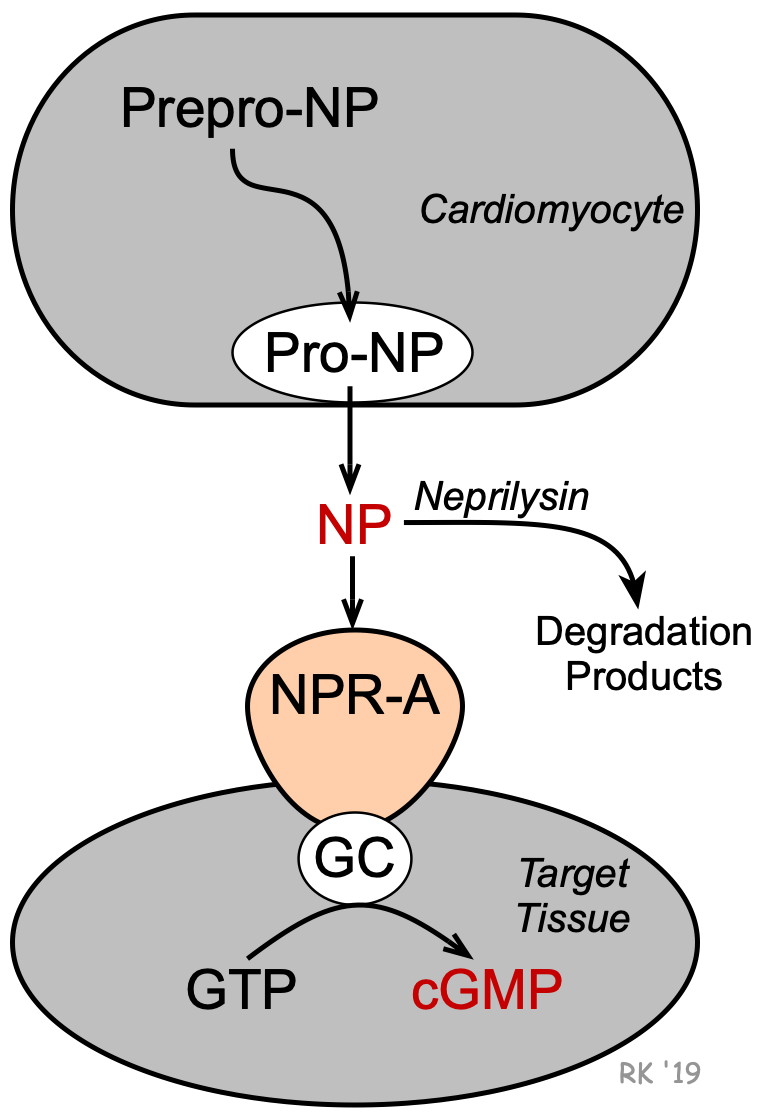 A second natriuretic peptide (brain-type natriuretic peptide; BNP) is a 32-amino acid peptide that is synthesized largely by the ventricles (as well as in the brain, where it was first identified). BNP is first synthesized as prepro-BNP, which is then cleaved to pro-BNP and finally to BNP. Like ANP, BNP is released by the same mechanisms that release ANP, and it has similar physiological actions. Proteolysis of pro-BNP (108 amino acids) results in BNP (32 amino acids) and the N-terminal piece of pro-BNP (NT-pro-BNP; 76 amino acids). Both BNP and NT-pro-BNP are sensitive, diagnostic markers for heart failure in patients.
A second natriuretic peptide (brain-type natriuretic peptide; BNP) is a 32-amino acid peptide that is synthesized largely by the ventricles (as well as in the brain, where it was first identified). BNP is first synthesized as prepro-BNP, which is then cleaved to pro-BNP and finally to BNP. Like ANP, BNP is released by the same mechanisms that release ANP, and it has similar physiological actions. Proteolysis of pro-BNP (108 amino acids) results in BNP (32 amino acids) and the N-terminal piece of pro-BNP (NT-pro-BNP; 76 amino acids). Both BNP and NT-pro-BNP are sensitive, diagnostic markers for heart failure in patients.
Circulating ANP and BNP can bind to natriuretic peptide receptor-A (NPR-A) in various tissues, particularly vascular and renal. NPR-A is coupled to particulate guanylyl cyclase (GC) that is associated with the NPR-A on the cellular membrane. GC activation leads to the formation of cGMP from GTP. The cGMP serves as a second messenger for the cellular actions of natriuretic peptides.
Neutral endopeptidase (NEP; also called neprilysin) is a circulating enzyme that degrades natriuretic peptides. Therefore, inhibition of this enzyme (e.g., by sacubitril) increases circulating levels of natriuretic peptides and potentiates their effects.
Cardiovascular and renal effects
Cardiovascular and Renal
Actions of Natriuretic Peptides
- Natriuresis
- Diuresis
- Improve glomerular filtration rate
& filtration fraction - Inhibit renin release
- ↓ circulating angiotensin II
- ↓ circulating aldosterone
- Systemic vasodilation
- Arterial hypotension
- Reduced venous pressure
- Reduced pulmonary capillary
wedge pressure
Natriuretic peptides (NPs) are involved in the long-term regulation of sodium and water balance, blood volume and arterial pressure. There are two major pathways of natriuretic peptide actions: 1) vasodilator effects, and 2) renal effects that lead to natriuresis and diuresis.
NPs directly dilate veins (increase venous compliance) and decrease central venous pressure, which reduces cardiac output by decreasing ventricular preload. NPs also dilate arteries, which decreases systemic vascular resistance and systemic arterial pressure. Chronic elevations of NPs appear to decrease arterial blood pressure primarily by decreasing systemic vascular resistance. The mechanism of systemic vasodilation involves NP receptor-mediated elevations in vascular smooth muscle cGMP and attenuation of sympathetic vascular tone. This latter mechanism may involve NPs acting on sites within the central nervous system and through inhibition of norepinephrine release by sympathetic nerve terminals.
NPs affect the kidneys by increasing glomerular filtration rate (GFR) and filtration fraction, which produces natriuresis (increased sodium excretion) and diuresis (increased fluid excretion). These renal effects of NPs are potassium sparing, unlike most diuretic drugs that are used to induce natriuresis and diuresis in patients.
A second renal action of NPs is that they decrease renin release, decreasing circulating levels of angiotensin II and aldosterone. This leads to further natriuresis and diuresis. Decreased angiotensin II also contributes to systemic vasodilation and decreased systemic vascular resistance.
Taken together, these actions of NPs decrease blood volume, arterial pressure, central venous pressure, pulmonary capillary wedge pressure, and cardiac output. To summarize, natriuretic peptides serve as a counter-regulatory system for the renin-angiotensin-aldosterone system (RAAS).
A recombinant human BNP, or nesiritide, was developed and evaluated for treating acute, decompensated congestive heart failure caused by systolic dysfunction. However, this drug is no longer used in the treatment of this condition because a large clinical trial failed improved clinical outcomes. A new class of drugs that are neutral endopeptidase (NEP, neprilysin) inhibitors (e.g., sacubitril), when combined with an angiotensin receptor blocker (ARB, valsartan) have been shown to be safe and effective in treating acute, decompensated heart failure. This drug combination blocks both angiotensin receptors (AT1) and the degradation of NPs, which enhances the RAAS counter-regulatory effects of NPs.
Revised 10/30/2023
 Cardiovascular Physiology Concepts, 3rd edition textbook, Published by Wolters Kluwer (2021)
Cardiovascular Physiology Concepts, 3rd edition textbook, Published by Wolters Kluwer (2021) Normal and Abnormal Blood Pressure, published by Richard E. Klabunde (2013)
Normal and Abnormal Blood Pressure, published by Richard E. Klabunde (2013)