Normal and Abnormal Electrical Conduction
Sequence of Cardiac Electrical Activation
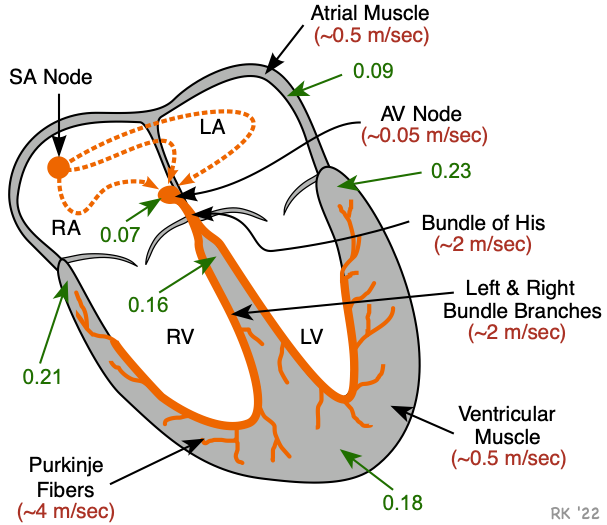 The action potentials generated by the SA node spread throughout the atria, primarily by cell-to-cell conduction at a velocity of about 0.5 m/sec (red number in figure). There is some functional evidence for the existence of specialized conducting pathways within the atria (termed internodal tracts). As the wave of action potentials depolarizes the atrial muscle, the cardiomyocytes contract by a process termed excitation-contraction coupling.
The action potentials generated by the SA node spread throughout the atria, primarily by cell-to-cell conduction at a velocity of about 0.5 m/sec (red number in figure). There is some functional evidence for the existence of specialized conducting pathways within the atria (termed internodal tracts). As the wave of action potentials depolarizes the atrial muscle, the cardiomyocytes contract by a process termed excitation-contraction coupling.
Normally, the only pathway available for action potentials to enter the ventricles is through a specialized region of cells (atrioventricular node, or AV node) located in the inferior-posterior region of the interatrial septum. The AV node is a highly specialized conducting tissue (cardiac, not neural) that slows the impulse conduction considerably (to about 0.05 m/sec), which allows for sufficient time to complete atrial depolarization and contraction before ventricular depolarization and contraction.
The impulses then enter the base of the ventricle at the Bundle of His and then follow the left and right bundle branches along the interventricular septum. These specialized fibers conduct the impulses at a very rapid velocity (about 2 m/sec). The bundle branches then divide into an extensive system of Purkinje fibers that conducts the impulses at high velocity (about 4 m/sec) throughout the ventricles. This results in rapid depolarization of ventricular myocytes throughout both ventricles.
The conduction system within the heart is vital because it permits a rapid and organized depolarization of ventricular myocytes that is necessary for the efficient generation of pressure during systole. The time (in seconds) to activate the different regions of the heart is shown in the figure (green arrows and numbers). Atrial activation is complete within about 0.09 sec (90 msec) following SA nodal firing. After a delay at the AV node, the septum becomes activated (0.16 sec). All the ventricular mass is activated by about 0.23 sec.
Regulation of Conduction
 The conduction of electrical impulses in the heart occurs cell-to-cell and highly depends on the rate of cell depolarization in both nodal and non-nodal cells. Therefore, because conduction velocity depends on the rate of depolarization, autonomic nerve activity, hormones, drugs and electrical changes in response to ischemia can alter the rate of depolarization and affect conduction velocity. Sympathetic activation increases conduction velocity in the AV node and in the His-Purkinje system by increasing the rate of depolarization (increasing slope of phase 0) of the action potentials. This leads to more rapid depolarization of adjacent cells, and more rapid conduction of action potentials (positive dromotropy). Sympathetic activation of the AV node reduces the normal delay of conduction through the AV node, reducing the time between atrial and ventricular contraction. The increase in AV nodal conduction velocity can be observed as a decrease in the PR interval of the electrocardiogram. Sympathetic stimulation of His-Purkinje pathways speeds up ventricular depolarization. Sympathetic nerves exert their actions on the AV node and His-Purkinje fibers by releasing the neurotransmitter norepinephrine that binds to beta-adrenoceptors, leading to an increase in intracellular cAMP. Therefore, drugs that block beta-adrenoceptors (beta-blockers) decrease conduction velocity.
The conduction of electrical impulses in the heart occurs cell-to-cell and highly depends on the rate of cell depolarization in both nodal and non-nodal cells. Therefore, because conduction velocity depends on the rate of depolarization, autonomic nerve activity, hormones, drugs and electrical changes in response to ischemia can alter the rate of depolarization and affect conduction velocity. Sympathetic activation increases conduction velocity in the AV node and in the His-Purkinje system by increasing the rate of depolarization (increasing slope of phase 0) of the action potentials. This leads to more rapid depolarization of adjacent cells, and more rapid conduction of action potentials (positive dromotropy). Sympathetic activation of the AV node reduces the normal delay of conduction through the AV node, reducing the time between atrial and ventricular contraction. The increase in AV nodal conduction velocity can be observed as a decrease in the PR interval of the electrocardiogram. Sympathetic stimulation of His-Purkinje pathways speeds up ventricular depolarization. Sympathetic nerves exert their actions on the AV node and His-Purkinje fibers by releasing the neurotransmitter norepinephrine that binds to beta-adrenoceptors, leading to an increase in intracellular cAMP. Therefore, drugs that block beta-adrenoceptors (beta-blockers) decrease conduction velocity.
Parasympathetic (vagal) activation decreases conduction velocity (negative dromotropy) at the AV node by decreasing the slope of phase 0 of the action potentials. This leads to slower depolarization of adjacent cells and reduced velocity of conduction. Acetylcholine, released by the vagus nerve, binds to cardiac muscarinic receptors, which decreases intracellular cAMP. Excessive vagal activation can produce AV block.
Calcium-channel blockers such as verapamil or diltiazem reduce the conduction velocity of impulses through the AV node and can produce AV block. These drugs inhibit the movement of calcium into the cell by blocking L-type calcium channels, which reduces the slope of phase 0. Antiarrhythmic drugs such as quinidine (a Class IA antiarrhythmic) that block fast sodium channels cause a decrease in conduction velocity in non-nodal tissue because the slope of phase 0 is primarily determined by fast sodium channels in non-nodal cells.
Myocardial ischemia and hypoxia reduce conduction velocity. In non-nodal cardiac tissue, cellular hypoxia leads to membrane depolarization, inactivation of fast sodium channels, a decrease in the slope of phase 0, and a decrease in action potential amplitude. These changes decrease the speed with which action potentials are conducted within the heart. This can have several consequences. First, activation of the heart will be delayed, and occasionally, the sequence of activation will be altered. This can seriously impair ventricular pressure development. Second, damage to the conducting system can precipitate tachyarrhythmias by reentry mechanisms. Click here to learn more about altered impulse conduction.
Revised 10/27/2023
 Cardiovascular Physiology Concepts, 3rd edition textbook, Published by Wolters Kluwer (2021)
Cardiovascular Physiology Concepts, 3rd edition textbook, Published by Wolters Kluwer (2021) Normal and Abnormal Blood Pressure, published by Richard E. Klabunde (2013)
Normal and Abnormal Blood Pressure, published by Richard E. Klabunde (2013)