Metabolic Mechanisms of Vasodilation
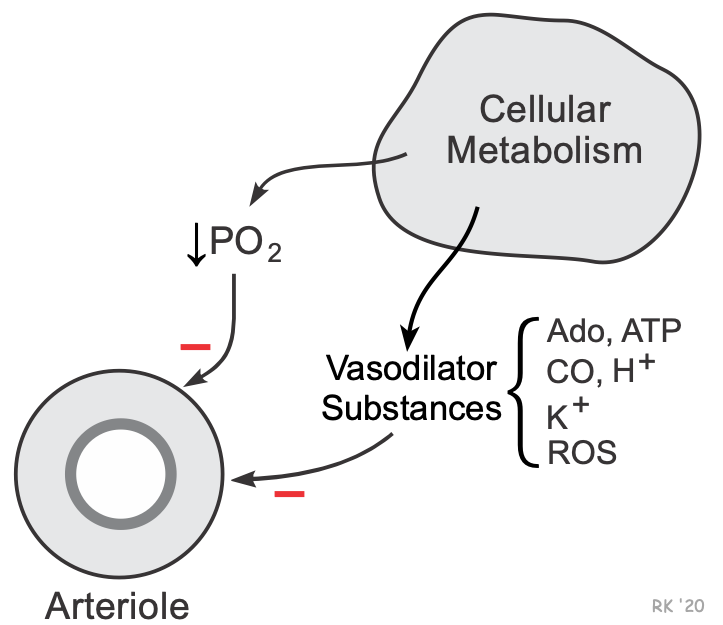 Blood flow is closely coupled to tissue metabolic activity in most organs of the body. For example, an increase in tissue metabolism, as occurs during muscle contraction or during changes in neuronal activity in the brain, leads to an increase in blood flow (active hyperemia). There is considerable evidence that actively metabolizing cells surrounding arterioles release vasoactive substances that cause vasodilation. This is termed the metabolic theory of blood flow regulation. Increases or decreases in metabolism lead to increases or decreases in the release of these vasodilator substances. These metabolic mechanisms ensure that the tissue is adequately supplied by oxygen and that products of metabolism (e.g., CO2, H+, lactate) are removed. Another mechanism that may couple blood flow and metabolism involves changes in the partial pressure of oxygen.
Blood flow is closely coupled to tissue metabolic activity in most organs of the body. For example, an increase in tissue metabolism, as occurs during muscle contraction or during changes in neuronal activity in the brain, leads to an increase in blood flow (active hyperemia). There is considerable evidence that actively metabolizing cells surrounding arterioles release vasoactive substances that cause vasodilation. This is termed the metabolic theory of blood flow regulation. Increases or decreases in metabolism lead to increases or decreases in the release of these vasodilator substances. These metabolic mechanisms ensure that the tissue is adequately supplied by oxygen and that products of metabolism (e.g., CO2, H+, lactate) are removed. Another mechanism that may couple blood flow and metabolism involves changes in the partial pressure of oxygen.
Several mechanisms that may be involved in the metabolic regulation of blood flow are summarized below:
Hypoxia:
Decreased tissue pO2 resulting from reduced oxygen supply or increased oxygen demand causes vasodilation. Hypoxia-induced vasodilation may be direct (inadequate O2 to sustain smooth muscle contraction) or indirect via the production of vasodilator metabolites. Note, however, that hypoxia induces vasoconstriction in the pulmonary circulation (i.e., hypoxic vasoconstriction), which likely involves the formation of reactive oxygen species, endothelin-1 or products of arachidonic acid metabolism.
Tissue Metabolites and Ions:
Adenosine is formed from cellular AMP acted upon by 5'-nucleotidase. The AMP is derived from the hydrolysis of intracellular ATP and ADP. Adenosine formation increases during hypoxia and increased oxygen consumption, especially if inadequate oxygen delivery accompanies the latter. Adenosine formation is an important mechanism for regulating coronary blood flow.
ATP, which is released by contracting skeletal muscle and by red blood cells, causes vasodilation by binding to purinergic receptors and by stimulating endothelial release of nitric oxide.
Potassium ion is released by contracting cardiac and skeletal muscle. Small increases in extracellular K+ produces hyperpolarization of vascular smooth muscle and relaxation through stimulation of the electrogenic Na+/K+-ATPase pump and increasing membrane conductance to K+ (K+ activated K+ channels). Extracellular K+ increases when there is an increase in action potential frequency because with each action potential, K+ leaves the cell. Normally, the Na+/K+-ATPase pump can restore the ionic gradients; however, the pump does not keep up with rapid depolarizations (i.e., there is a time lag) during muscle contractions and this causes K+ to accumulate in the extracellular space. Potassium ion appears to play a significant role in causing active hyperemia in contracting skeletal muscle.
Carbon dioxide formation increases during states of increased oxidative metabolism. It readily diffuses from parenchymal cells in which it is produced to the vascular smooth muscle of blood vessels, where it causes vasodilation. CO2 plays a significant role in regulating cerebral blood flow.
Hydrogen ion increases when CO2 increases or during states of increased anaerobic metabolism, which can produce metabolic acidosis. Like CO2, increased H+ (decreased pH) causes vasodilation, particularly in the cerebral circulation.
Lactic acid, a product of anaerobic metabolism, is a vasodilator because of its pH-lowering effect.
Inorganic phosphate is released by the hydrolysis of adenine nucleotides. It may have some vasodilatory activity in contracting skeletal muscle.
Osmolality of the blood is increased, for example, in contracting skeletal muscle. This nonspecific increase in osmolality leads to vascular smooth muscle relaxation and vasodilation.
Reactive oxygen species such as superoxide anion and its product, hydrogen peroxide, are elevated by increased tissue metabolism and may contribute to vasodilation. They play a prominent role in vasodilation associated with inflammation.
Revised 12/31/2022
 Cardiovascular Physiology Concepts, 3rd edition textbook, Published by Wolters Kluwer (2021)
Cardiovascular Physiology Concepts, 3rd edition textbook, Published by Wolters Kluwer (2021) Normal and Abnormal Blood Pressure, published by Richard E. Klabunde (2013)
Normal and Abnormal Blood Pressure, published by Richard E. Klabunde (2013)